Amyotrophic lateral sclerosis (ALS) and frontotemporal dementia are associated with the aggregation of certain RNA-binding proteins that are part of stress granules, including TAR DNA-binding protein 43 (TDP-43), Fused in Sarcoma (FUS) and Heterogeneous nuclear ribonucleoprotein A (hnRNPA). Stress granules are cytoplasmic membraneless organelles that are formed in response to stresses that halt protein production. These organelles collect the mRNA that was being translated, possibly to protect the RNA or to ensure that no damaged proteins are produced.
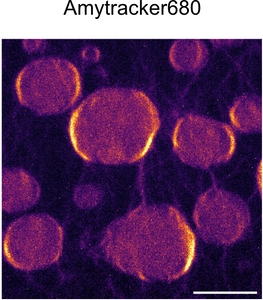
A study by Morelli et al. published in Nature Chemistry investigates how interactions with RNA modulate the aggregation of hnRNPA1A. Using an in vitro model around recombinant hnRNPA1A, the researchers show that this protein can phase-separate into molecular condensates where it then aggregates over time. Using Amytracker 680 to visualise hnRNPA1A aggregation, the authors could confirm that the amyloid fibrils are mainly found at the condensates’ surface.
Adding various concentrations of RNA into the system changes the dynamics of the condensation-to-aggregation process: In a system without RNA, hnRNPA1A condensates and progressively forms amyloid-like aggregates. However, this process is slow and does not reach a plateau in the timeline that was explored in the study. When RNA is added, hnRNPA1A aggregates faster, reaching a plateau before the end of the experiment and high amounts of RNA prevent condensation, but not aggregation, and hnRNPA1A forms aggregates directly.
Image: Confocal microscopy of hnRNPA1A condensates stained with Amytracker. reveal elevated fluorescence on the condensate surface indication transition from condenstation to aggregation of hnRNPA1A. Scale bar: 5 µm. Image from Figure S11 by Morelli, C., Faltova, L., Capasso Palmiero, U. et al. (2024) Nature Chemistry, 16(7), 1052-1061 (CC BY 4.0).
